Retro Ski Day 2024
Wie schon im letzten Jahr wurde vom Tiroler Krebsforschungsinstitut (TKFI) für seine ForscherInnen und KollaborationspartnerInnen ein “Retro Ski…
Bei der Onkogenese verändern sich normale Zellen im Körper und werden zu Tumorzellen, die sich in weiterer Folge unkontrolliert vermehren und zentrale Körperfunktionen beeinträchtigen können. Dafür sind mitunter Veränderungen im Erbgut (Mutationen) oder Fehler beim Ablesen der Erbinformation verantwortlich.
Die ForscherInnen am Tiroler Krebsforschungsinstitut (TKFI)
untersuchen die molekularen Ursachen der Entstehung von
Krebserkrankungen. Arbeitsgruppen beider Innsbrucker
Universitäten (Medizinische Universität Innsbruck (MUI) und Leopold-Franzens-Universität (LFU)) versuchen am TKFI die pathologischen Auswirkungen unterschiedlicher Cancer Driver zu verstehen und deren Wirkungsweise auf molekularer Ebene zu ergründen.
Unsere Vision ist es, neue Ansatzpunkte für personalisierte
Krebstherapien zu identifizieren.
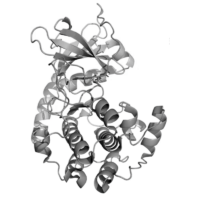
Die gelisteten Projekte beschäftigen sich mit den molekularen Ursachen für die Entstehung und das Voranschreiten von Krebserkrankungen.
Der Fokus der involvierten Projekte liegt auf der Krebstherapie und Mechanismen der Medikamentenresistenz.
Die assoziierten Projekte behandeln unter anderem Themen wie die Analytik von Atemgas, die Dechiffrierung von Onkoproteinen und die Etablierung von Organoiden.
Hier listen wir chronologisch Neuigkeiten und wissenschaftlichen Errungenschaften für Sie auf.
Wie schon im letzten Jahr wurde vom Tiroler Krebsforschungsinstitut (TKFI) für seine ForscherInnen und KollaborationspartnerInnen ein “Retro Ski…
Im Rahmen der nationalen Initiative „Unser Einsatz für Österreich“ von Casinos Austria AG (CASAG) unterstreicht das Casino Innsbruck…